At first glance, the new class of drugs appeared to be potent weapons in the fight against type 2 diabetes. Known as CB1 receptor antagonists, the drugs block the action of a cellular receptor called CB1, essentially helping modulate how a person experiences hunger, pain, and other sensations. Blocking that receptor, researchers hoped, could yield effective treatments for both obesity and diabetes. But subsequent studies also found a potential link to psychiatric side effects including anxiety and suicidal thoughts. Although the efficacy of the CB1 class in treating diabetes was not in doubt, the potential side effects were considered unacceptable. The result: one CB1 receptor antagonist, rimonabant, was withdrawn from the market, and work on developing others stalled.
The story, however, does not end there. More recently, researchers have discovered genetic variations that could identify those patients most susceptible to the side effects. The upshot: the CB1 class may yet emerge as a viable treatment option in combating diabetes, with genetic testing available to identify those patients who would suffer the most from its potential side effects.
The saga of the CB1 receptor antagonists illustrates the emergence of biomarkers as a powerful driver of progress in the diabetes field. Biomarkers—genes, proteins, or other measurable indicators—can be used to monitor key biological conditions including disease states, to identify those patients most likely to benefit from a therapy, or to spot those with a high likelihood of experiencing side effects from a particular drug. And while, for now, hemoglobin A1c (HbA1c) remains the only well-proven diabetes biomarker, that is likely to change.
The Boston Consulting Group has just concluded an exhaustive four-month study on the future of diabetes care across all health-care sectors, including an analysis of how the increase in cases of the disease will impact pharmaceutical companies, medical-device manufacturers, payers, and providers. Although no transformational drug therapies may be on the horizon yet, we believe that the current aggressive push to uncover and validate new biomarkers will transform diabetes treatment in the long term. It will lead to the development of new tools to help physicians stratify patients on the basis of their susceptibility to therapies’ side effects and their likely response to particular drugs. Biomarkers are also expected to help identify individuals with prediabetic conditions, those at risk of rapid disease progression, and those at high risk of experiencing complications.
The Diabetes Threat Grows as a Cure Remains Elusive
Consider that by 2030, 1.6 percent of U.S. GDP—about 9.6 percent of spending on U.S. health care overall—will be consumed by diabetes. In countries with an even higher incidence of the disease, the toll will be greater still. The United Arab Emirates is on track to see 13.1 percent of its health-care budget going to diabetes care by 2030, given the fast increase of diabetes risk factors such as obesity in its growing population.
The devastation wrought by the diabetes epidemic is difficult to overstate. In the United States, it is estimated that 9 percent of the population suffers from the disease today, while in China the share is 5 percent. The trajectory is just as frightening: by 2030, 12 percent of Americans and 6 percent of people in China are projected to have diabetes. And many of those individuals afflicted with diabetes are undiagnosed—an estimated 25 percent of the total number of people with the disease in the U.S. and up to 70 percent of type 2 diabetes sufferers in some rapidly developing economies.
The forces driving these increasing numbers are, unfortunately, difficult to influence. The most prominent: an expanding—and aging—global population, along with the adoption of lifestyles that are based on a Western diet and limited exercise, pushing obesity rates upward.
At the same time, a treatment that can stop—or even reverse—diabetes has not been developed. Marketed therapies primarily aim to lower blood glucose levels as monitored by HbA1c. Pre-insulin drugs such as metformin and sulfonylureas work in this way, as do more recent therapies such as DPP-4 inhibitors, GLP-1, and SGLT2 inhibitors.
And while these novel noninsulin antidiabetic drugs have clear benefits—namely, weight loss and better glucose control—all come at several times the price of current insulin therapies. For this reason, many health-care systems are debating the value of the drugs in health-economics terms and are putting up higher hurdles for the evidence that their manufacturers need to provide. A
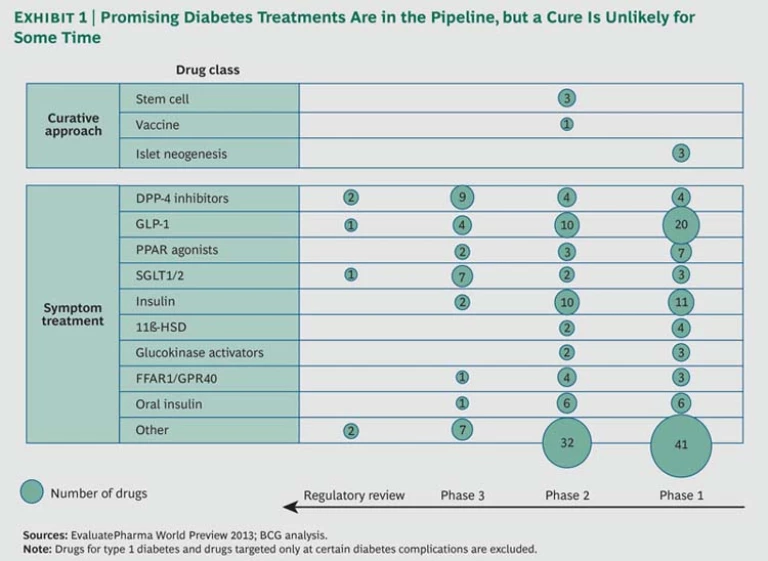
Certainly, there are promising new diabetes treatments in the pipeline, including oral versions of insulin and GLP-1 formulations, which spare patients the need for injections. But it appears that none hold the promise of a cure, and most are likely to face the same cost-benefit challenges. (See Exhibit 1.)
The work on biomarkers is critical to address the crippling burden—both financial and personal—of diabetes, and also to accelerate the development of new, more targeted therapies.
And companies will not have to wait decades to see the impact of biomarker research on real-life clinical practice. The benefits of this work will be tangible in a matter of years. Just recently, a new biomarker for diabetes was discovered: 2-aminoadipic acid (2-AAA), which is claimed to be able to predict a person’s propensity to develop diabetes more effectively than the risk factors we observe today, such as obesity. Researchers expect this biomarker to be the first of many to come.
Complexity Impedes Progress
Powerful biomarkers have emerged in other fields and contributed enormously to therapeutic advancement. The breast cancer drug Herceptin, for example, is effective in cancers where the HER2 gene is overexpressed. Today, HER2 testing is widespread to identify those patients who are likely to respond favorably to the drug.
In diabetes, however, HbA1c, discovered more than 50 years ago, has remained the sole proven biomarker. A number of factors have made progress in the field of diabetes biomarkers particularly challenging. For one thing, the disease is complex, involving numerous biological processes—or pathways—in the body. For another, a variety of patient-specific factors such as age, weight, and cardiovascular health have a large impact on whether the disease develops and how it progresses.
At the same time, there is increasing evidence that diabetes is not simply two disease states, type 1 and type 2, but rather a continuum of illnesses. In some cases, the disease manifests itself early and with only limited impact from factors such as weight gain. In others, the disease emerges after an event such as gestational diabetes. And in still other cases, patients have major risk factors such as being significantly overweight throughout much of their life but do not develop diabetes until much later than might be expected.
As a result, diabetic patients may differ dramatically in terms of the speed of disease progression, the likelihood that they will experience serious complications, and their response to various treatments.
The Way Forward
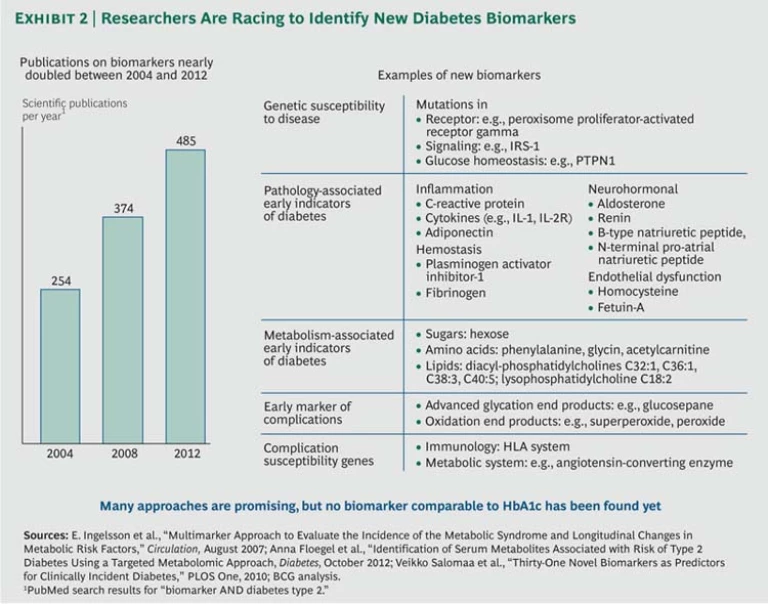
Now, however, progress in genomics, along with technological advances that allow researchers to harness massive amounts of patient data involving multiple genes and other risk factors such as age, body mass index, and drug usage, are making it possible to find critical associations between genetic variation and the disease. The result has been a sharp increase in biomarker research, with the number of scientific papers on the subject almost doubling from 254 to 485 between 2004 and 2012. (See Exhibit 2.)
And that progress has not gone unnoticed by the venture capital community, where there is increased interest in diabetes biomarker work. Venture capital firms, for example, are among the investors in VeraLight, developer of the SCOUT DS technology, a noninvasive screening system for prediabetes and type 2 diabetes based on biomarkers found in the skin. The technology was purchased in April 2013 by medical diagnostic company Miraculins. And Tethys Bioscience, which developed a biomarker-based test to identify patients at risk for type 2 diabetes, has raised a total of $103 million—much of it from the venture capital community.
The result will be not one comprehensive biomarker for the disease but rather multiple biomarkers that will create new ways of identifying high-risk patients early and targeting therapies to match a patient’s particular pathology. The biomarkers are likely to provide insight in five key areas of diabetes treatment.
Identifying Patients Likely to See Rapid Disease Progression. The degree to which aggressive treatment is necessary in a patient with diabetes depends in part on how rapidly the disease will develop over time. The identification of these patients will become easier as well-known risk factors including age and body mass index are assessed in combination with biomarkers such as C-reactive protein—a measure of inflammation—and new biomarkers yet to be discovered. While the link between inflammation and cardiovascular disease has been well established, more recent research has expanded the perspective to the link between inflammation and diabetes.
Other biomarkers are being studied that may provide insight into how patients’ genetic variations may impact how sensitive they are to insulin and glucagon in the body. Insulin and glucagon work in opposing ways, with the former triggering the removal of glucose from the bloodstream and the latter triggering the release of glucose from reserves in the body. The degree of sensitivity to both peptides will affect the body’s ability to regulate blood glucose levels effectively and can predict patients’ receptivity to new therapies.
Spotting Patients with a High Likelihood of Complications. Diabetes complications such as nephropathy, retinopathy, and neuropathy, which stem from excessively high levels of glucose in the blood, are the single largest component of diabetes costs overall in developed countries. But individuals differ in how sensitive their cells are to glucose. Biomarkers to identify people who are less able to tolerate elevated glucose levels could predict those with a high likelihood of developing complications. A number of biomarkers are being studied in this area, including advanced glycation end products (AGEs)—proteins or lipids that are altered after exposure to sugars and have been linked to diabetes complications. Once those patients with a high risk of complications are identified, they can be treated at an earlier stage with either noninsulin antidiabetic therapies or insulin, as well as with more frequent blood glucose monitoring and a restricted diet.
Matching Patients with the Treatments to Which They Are Most Likely to Respond. Companies are under increasing pressure to prove that their drugs are adding therapeutic value at the increased cost they usually command. Better targeting of therapies to those patients who are likely to respond can help deliver a compelling cost-benefit proposition. Consider the FFAR1/GPR40 agonist class of drugs in the pipeline—compounds that stimulate the action of a cell receptor called FFAR1 or GPR40, which is linked to insulin secretion. Research has uncovered a genetic variation in the receptor in some people that leads to differences in how efficacious these drugs may be.
Identifying Patients Who Are Susceptible to Side Effects. The tradeoff between risks and benefits will become easier to discern as biomarkers emerge that reveal which patients are likely to suffer serious adverse reactions to certain drugs. That insight will be invaluable and could help address concerns such as those raised by regulators over a possible link between newer noninsulin antidiabetic drugs and cancer.
Identifying and Validating Targets. Biomarkers are also likely to play a central role in developing diabetes drugs with novel modes of action. The new class of drugs known as SGLT2 inhibitors grew out of research studying people with a genetic mutation that leads to a condition called familial renal glycosuria. In particular, biomarkers related to obesity are likely to command significant focus and to yield new targets addressing both obesity and diabetes treatments, owing to the strong link between the two conditions. Studies have shown that weight loss after procedures such as gastric bypass surgery can lead to a significant reduction in diabetes severity.
Tailored Treatment
Different combinations of all five areas of biomarker advances will be brought to bear on treatment decisions. Identifying patients most at risk—through either rapid disease progression or likelihood of suffering complications—will help target treatment more effectively. And biomarker tools will also help match the optimal regimen of therapies to each patient on the basis of an assessment of likely effectiveness and side effects.
This integrated approach is critical, because governments and payers may find they are unable to bear the cost of treating every patient in the future with current therapies. That would break the bank.