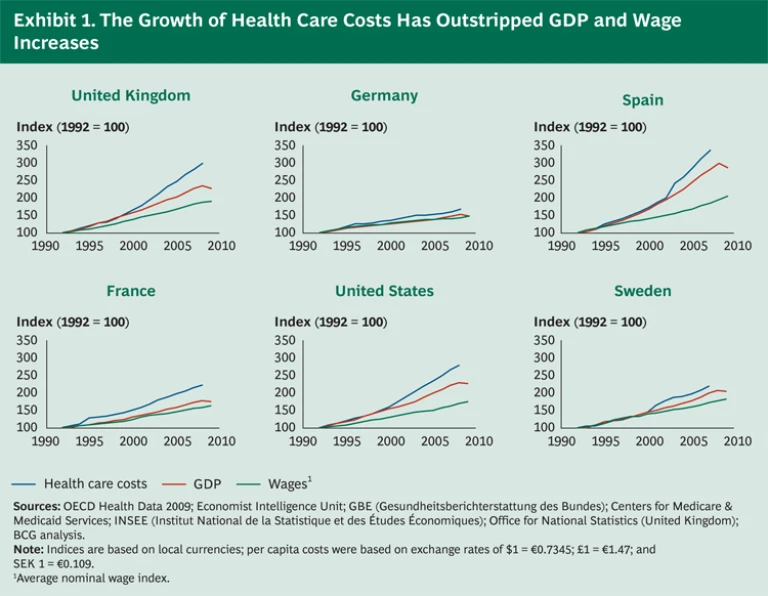
Health care costs continue to rise at an alarming rate, far outpacing the growth of both national economies and household incomes. In the United States, the bill stands at 18 percent of GDP—and costs have risen three times faster than economic growth over the past 20 years. Other major Western countries face similar challenges, with health care costs consuming nearly 10 percent of GDP and increasing at twice the rate of economic growth. (See Exhibit 1.) Aging populations—the “grey tsunami”—will only lead to greater demand for medical care, while unprecedented pension obligations pressure national treasuries already severely strained by the global economic downturn.
The answer, one might think, is simple: drive out inefficiencies, slash reimbursement, control margins, and limit utilization. But a focus on volume and cost alone is misguided and counterproductive. For two decades, governments and other payers around the world have applied these levers—yet health care costs continue their inexorable ascent. And there seem to be few viable alternatives: cutting benefits inevitably leads to charges of rationing, while increasing taxes on all but the wealthiest is dismissed as politically untenable.
A new approach is needed—one that will reorient health care systems around a more encompassing metric. Four years ago, Michael Porter and Elizabeth Teisberg introduced just such a concept—value-based health care (VBHC)—in their book, Redefining Health Care: Creating Value-Based Competition on Results (Harvard Business Press, 2006). The goal of VBHC is not to minimize costs but to maximize “value,” defined as patient outcomes divided by costs. To implement VBHC, providers and payers must identify, codify, and promote treatment protocols that are proven to yield better, more cost-effective care. The patient’s well-being is thus an explicit part of the equation.
Some health care experts are quick to dismiss VBHC on the grounds that a lack of timely and reliable data, the divergent interests of stakeholders, and the industry’s aversion to wholesale transformation would stonewall meaningful progress. In the real world, the argument goes, VBHC would be impossible to orchestrate. But the experience of the Swedish health care system suggests otherwise. Over the past several decades, Sweden’s health care sector has developed a number of disease registries—vast repositories of data on the outcomes for different patient groups. The information has helped providers identify value-based treatment protocols, leading to improvements in both efficiency and patient outcomes.
Sweden has not created a full-scale version of VBHC, but it has found a practical solution to the single largest challenge—obtaining timely and reliable data. This approach provides a glimpse of what other industrialized societies could achieve by investing in disease registries and collecting outcomes data at a national or even international level.
The Unsustainable Status Quo
Rising health care costs would be a serious problem even if improvements in health care quality had kept pace. Unfortunately, they have not. In the United States, for example, a survey by the Agency for Healthcare Research and Quality found that while health care expenditures doubled between 1994 and 2005, the quality of care—measured in terms of effectiveness, patient safety, timeliness, and patient-centeredness—improved by barely a third. Moreover, while U.S. health care spending per capita vastly exceeds that of any other country, Americans have fewer healthy life years—a measure of how long they live before health problems have a lasting impact on activity—than citizens of most other advanced economies.
Some might argue that the U.S. results are a product of a more heterogeneous population and greater income disparity. But even among discrete groups of well-off Americans, health care outcomes are, at best, on a par with the basic standard of care in other countries. In 2008, for example, the wealthiest 10 percent of Americans had the same life expectancy (81 years) as the average Swede, despite the fact that per capita health care costs in the United States are close to twice as high as in Sweden.
Why is this happening? Around 70 percent of overall health care costs are incurred at the provider level—in hospitals and ambulatory surgery units, physicians’ offices, and rehab centers.
While these problems are ubiquitous across health systems in the OECD nations, the U.S. system illustrates them most vividly. With hospitals and physicians paid primarily on the basis of a per-procedure model, they gravitate toward the most profitable procedures, driving up those volumes. Similarly, reimbursement structures encourage drug and device companies to seek the highest available price per treatment and the maximum number of customers. This provides the impetus for product innovation—but the system also relies heavily on targeted marketing, physician detailing (sales and promotion visits), and direct-to-consumer advertising to stimulate demand. Critics argue that such efforts to stoke demand exploit a core flaw in the U.S. system: because the buying decision is often made by the physician or patient rather than the payer, there is no natural brake on demand. Meanwhile, the discussion of value is all but lost.
The problem goes right to the top. The journal Health Affairs asked the chairpersons of more than 700 U.S. hospitals to name their board’s top two priorities. Roughly half did not name quality of care.
What is required is a system that will bring costs and outcomes into equilibrium. Porter and Teisberg’s argument for VBHC is founded on a core belief that providers should organize care around the patient and his or her disease. By analyzing treatment outcomes, providers can identify, codify, and disseminate best practices. Payers can then offer incentives to propagate the “right” behavior and practices across the system.
VBHC echoes the practice of disease management, which optimizes patient outcomes by focusing on the critical moments in the evolution of a disease. Developed by BCG’s Dave Matheson in the late 1980s, disease management has been used to manage chronic disorders such as diabetes and congestive heart failure. It is based on the premise that better, more cost-effective care requires a system-based view of health care delivery. Like VBHC, it marks a sharp departure from traditional “component management,” which focuses on lowering unit costs and restricting utilization. Disease management recognizes that each chronic disease has a discernible economic structure, which can be predicted and managed to reduce complications, slow the disease’s progression, and improve outcomes.
Disease management is a total quality management (TQM) program. Providers educate patients on prevention and emphasize coaching over direct intervention. When interventions are necessary, they are done in a timely and cost-efficient manner, making use of the right treatment paradigm for a given condition. Most important, the patient’s medical condition, clinician encounters, and cost of treatment are tracked throughout the process, providing a trove of valuable information that can be used to refine treatment protocols.
This approach has been used around the world, but it remains far from widespread. Payers often lack the data infrastructure required to track diagnoses, treatments, outcomes, and costs at the patient level. This makes it almost impossible to identify best-practice treatments, let alone disseminate them throughout a health care system. The absence of a coordinated approach has given rise to an extremely broad array of treatment protocols. “As we look across the enormous differences in health care spending in our country,” wrote Atul Gawande, a U.S.-based surgeon and best-selling author, “what we are witnessing are enormous differences in the way medicine is practiced.”
VBHC expands on the basic principles of disease management by offering a prism through which to evaluate quality across a wide variety of medical conditions. Like disease management, its success hinges on the aggregation and analysis of treatment and outcomes data. VBHC, however, focuses less on developing prescriptive protocols and guidelines; and it leaves more room for novel approaches to clinical practice, enabling breakthroughs in treatment (and value delivered) over time.
Sweden’s approach to collecting data—patient-level diagnoses, treatments, and clinical results—offers a glimpse into the art of the possible. Its decades-old system of disease registries provides the backbone of this information architecture. Sweden’s emphasis on tracking treatments and outcomes began in the early 1800s, when maternity nurses were required to report key metrics on delivery outcomes to the local doctor. The physician, in turn, would follow up with the nurses who had posted the worst results, seeking explanations and providing guidance. More modern registries covering a broader array of conditions emerged in the early 1970s.
Sweden now boasts more than 70 registries that cover about 25 percent of national health expenditures. About a third of these registries collect patient data on more than 85 percent of all Swedish patients diagnosed with a given condition—and many have been in place long enough to provide a longitudinal view of the evolving patterns of practice and outcomes. Comprehensive registries tend to lead to more powerful insights into outcomes.
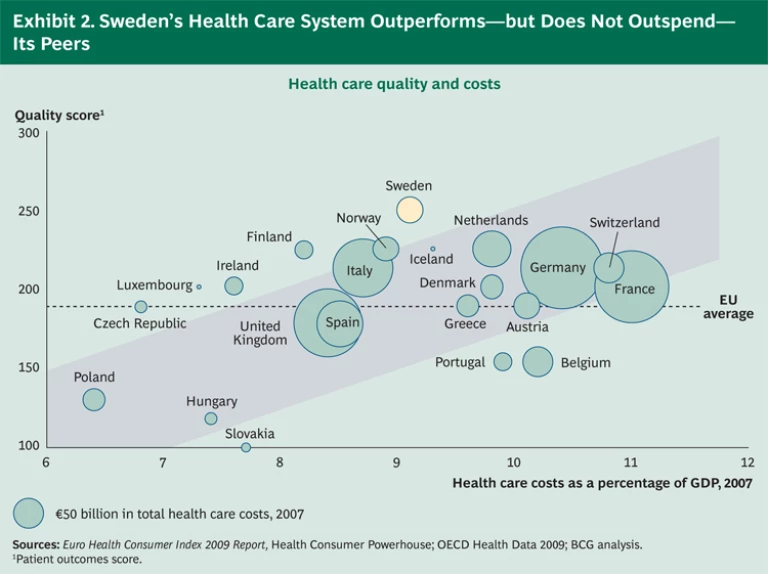
There is growing recognition that registries have contributed to the success of the country’s health care system. A recent study found Sweden to have the best health care outcomes in Europe, even though its health care costs, as a percentage of GDP, hover around the European average. (See Exhibit 2.) More specifically, the registries have been instrumental in providing benchmarks, defining best-practice protocols, and recalibrating incentives so that insights into treatments and outcomes find their way into clinical practice. (See the sidebar “Leveraging Data to Improve the Treatment of Acute Lymphoblastic Leukemia (ALL).”)
Recognizing the link between data and health care quality, the Swedish government doubled its financial support for the registries in December 2009 and is considering significant investments to further promote VBHC. The ongoing support for registries has as much to do with their quality as with their coverage. Most registries were initiated by the relevant medical-specialist societies, which continue to play a leading role in defining metrics and validating data. As a result, physicians are highly committed to the system. For example, the Swedish National Diabetes Register draws data from 95 percent of all departments of medicine and about 75 percent of all primary-care centers.
This level of involvement has created a system in which tracking outcomes and sharing best practices—“important information from dearly bought experiences,” in the words of one Swedish health professional—are rapidly becoming standard practice. It has enabled in-depth research on outcomes and has allowed protocols to coalesce around proven, highly effective treatments, with impressive results across medical interventions ranging from surgery to pharmaceutical therapies.
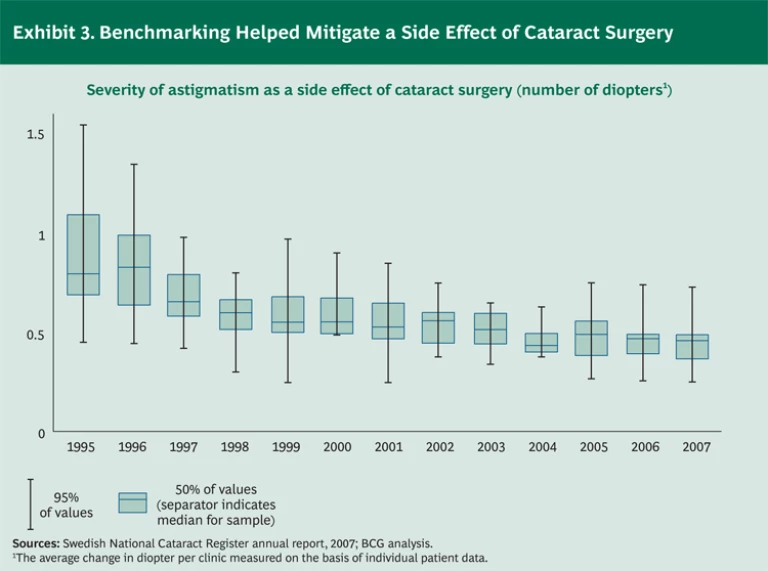
Although we did not compare Sweden’s outcomes with those of other countries, we did find several examples in which the link between registries, research, and protocols has had a clear and measurable impact on patients’ well-being. Sweden’s National Cataract Register is a case in point. Fifteen years ago, astigmatism of at least 0.68 diopter was a normal side effect of cataract surgery. (See Exhibit 3.) By 2007, the median astigmatism had fallen by one-third, to 0.45 diopter. More important, the variability in outcomes had decreased by 56 percent—and the risk of astigmatism greater than 0.50 diopter, which would often require corrective glasses, had been virtually eliminated. This improvement led to higher patient satisfaction, fewer corrective procedures, and lower costs. The adoption of improved operating techniques, which was a major factor behind the enhanced outcomes in cataract surgery, was facilitated and accelerated by the transparency created by the registry.
Building Effective Registries
The value of registries is determined by the volume and quality of the data. The more records that the registries contain, the greater is the chance that researchers can isolate significant patterns, such as important differences among patient subpopulations. These patterns, in turn, can indicate the success or failure of particular treatments. Ideally, registries should be national or multinational in scope.
Unfortunately, efforts to build large-scale international registries have had disappointing results. Projects sponsored by the Commonwealth Fund and the OECD have proceeded slowly and produced only limited agreement on the metrics used to benchmark outcomes. At the national level, however, registries are taking steps to broaden their horizons. Data from almost half of Sweden’s registries are being used in other countries, particularly the Nordic countries. In addition, some registries are formalizing arrangements to foster international collaboration. For example, the Swedish Spine Register, Schön Kliniken in Germany, and Global Health Partner of Sweden recently agreed to pool their data on spinal surgery outcomes.
The almost entrepreneurial expansion of individual registries underscores a critical factor in their success: medical and clinical professionals can and should take the lead in their development and maintenance. Specialty associations, in particular, must take an active role in establishing registries to ensure that the metrics used to benchmark outcomes are sound and scientifically valid. A lack of support from physicians can severely compromise a registry’s effectiveness. (Click “Registries Require Physician Support,” below.)
Registries Require Physician Support
In 2000, an outcome-measurement system was established in Germany called Bundesgeschäftsstelle Qualitätssicherung (BQS). The system, which was initiated by the national hospital and health plan associations, published its first report in 2001. It currently reports on 22 surgical procedures, representing 22 percent of all hospital cases.
Data collection for BQS is mandatory for all German hospitals, but since doctors were not sufficiently involved in its development, they never fully supported the approach. Many claim that the process for collecting data is inconsistent and that the quality of data is poor. The lack of support is evident in the rigor (or lack thereof) of the data. When a recent scientific study reviewed the intra- and postoperative complications of cholecystectomy captured in the BQS registry it found that less than half of the reviewed cases were correctly documented.
Similarly, the leadership of the Swedish Association of General Practice (SFAM) has been skeptical about the value of outcomes registries in their discipline. As a result, with the exception of diabetes, general practice remains one of the few areas of Swedish medicine that thus far lack outcomes registries.
Clinicians also need to feel a sense of ownership of the registries. After all, it’s up to them to supply the information, investigate the data, and apply their findings. This cannot be just another bureaucratic obligation, nor should it be cumbersome. The system should provide both the technology to make reporting seem routine and the analytical tools to enable users to benchmark clinical results and spot patterns. More advanced systems can provide doctors with the granular information they need to support clinical decision-making—for example, by enabling them to adjust treatments for specific patient subgroups. (An example of good decision-making support is described in the sidebar “The Impact of the Swedish Rheumatoid Arthritis Registry,” below.)
The Impact of the Swedish Rheumatoid Arthritis Registry
The Swedish Rheumatoid Arthritis Registry exemplifies best practice. All of Sweden’s rheumatoid arthritis clinics report to the registry. But the coverage varies by region and is influenced in part by the proportion of patients who are treated by specialists. Nationally, the registry covers about 60 percent of all rheumatoid arthritis patients, although in some regions the coverage exceeds 80 percent. The registry records prescribed treatments and outcomes, along with each patient’s perceived health status and use of drugs. The quality and depth of the data have provided a foundation for internationally recognized academic research and cooperative projects involving government and commercial agencies.
Data from the registry, combined with data from national population databases, produced a health-economic analysis of the effect of TNF-α inhibitors, a new class of drugs introduced during the past decade to fight autoimmune disorders. This analysis, in turn, enabled the value-based reimbursement of those drugs and allowed physicians to tailor their treatment protocols to individual patients, making personalized medicine a reality. For example, data showed that smokers respond to the treatment at one-quarter the rate of nonsmokers. As a result, some Swedish clinicians require patients to enroll in a smoking-cessation program as a prerequisite to treatment with TNF-α inhibitors.
These inhibitors are now considered a cost-efficient way of treating rheumatoid arthritis. But an apparent severe side effect could have prevented their adoption. There was some initial evidence that the inhibitors, by weakening the immune system, could triple the risk of lymphoma relative to the general population. Data from the registry, however, showed that all new rheumatology patients had an increased risk for lymphoma, regardless of whether they were treated with the inhibitor. Thus, the registry-based analysis presented to the European Medical Association in Brussels prevented the drug from being pulled from the market.
In Sweden, specialist societies or associations have been able to develop effective, comprehensive registries by focusing on the following four building blocks:
- Data Capture. To have a material impact on care, data must be collected from a high proportion of cases using a standardized format and set of metrics, and data collection needs to become part of daily clinical practice. Ideally, data should be captured automatically and integrated into electronic medical records, but so far this has been the exception rather than the rule. In Sweden, for instance, data are gathered using a broad range of platforms, including some that are not yet Web-based.
- Data Use. The data should be available to interdisciplinary groups, including epidemiologists, statisticians, and clinical experts. These experts will establish benchmarks for clinical performance, determine the reasons for variations in outcomes, and suggest best-practice and improved clinical guidelines. Where possible, the data should be analyzed against the backdrop of other databases such as epidemiological registries or biobanks.
- Infrastructure. Information systems (IS) and information technology (IT) need to facilitate data capture, access, and analysis. The infrastructure must be designed to identify and work through issues concerning the compatibility of data to ensure that it captures as many cases as possible. Registries generally do not need a substantial start-up investment, but they do require significant long-term investments in order to keep complexity in check, maintain shared IS and IT platforms, and ensure data compatibility.
- Governance. The integrity and privacy of patient information are paramount—but the impact of registries hinges on the data being available to a range of stakeholders, including not just providers but also commercial entities, which can use the data to steer the development of new products (and make it easier to assess their impact). A clear legal framework is critical to striking a balance between these sometimes competing interests.
Quantifying the Cost and Impact of Sweden's Registries
Although the anecdotal evidence of the power of registries is compelling, it can be difficult to isolate and quantify their benefits beyond looking at the relative performance of the overall health care system. Nonetheless, Sweden’s disease registries have become an integral part of that country’s health care system and have had a demonstrable impact on the practices of a range of stakeholders.
For example, Swedish payers, such as the Stockholm County Council, are introducing outcomes-based reimbursement for health care providers, while private providers are increasingly required to report their cases to the registries to ensure that payers get value for their money. As a result, some providers are actively seeking opportunities to collaborate with drug and device companies that are able to demonstrate how their products and services lead to better outcomes.
A value-based approach is also working its way into incentives for other stakeholders. For example, Sweden’s Dental and Pharmaceutical Benefits Agency (TLV) has for some years set reimbursement levels for drugs on the basis of their health-economic value—basically, their impact on patients and society. The next development is to link this assessment to the registries. Similarly, a health-economic analysis of TNF-α inhibitors, which are used to treat autoimmune disorders, is currently under way. It will compare the performance of different members of this drug class on the basis of data from outcomes registries and population registries (which would show phenomena such as absenteeism).
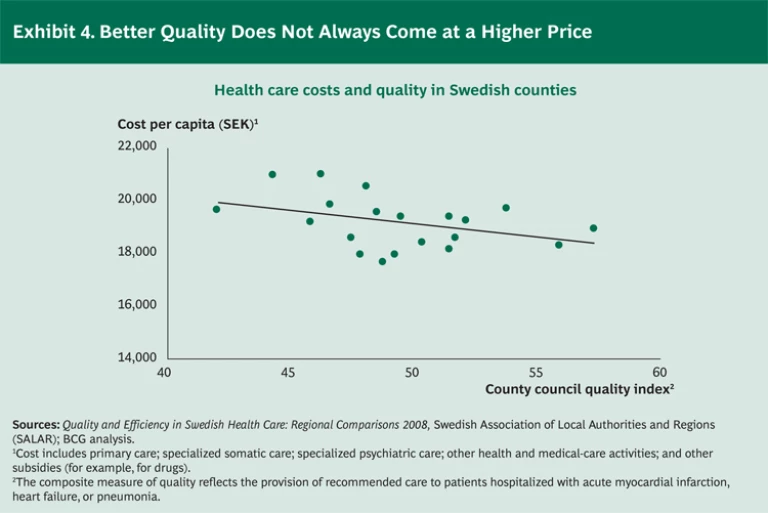
It’s important to note that on a systemwide level, high-quality care need not break the bank. In fact, our analysis of Swedish health regions demonstrates the opposite effect: regions with higher-quality care generate lower health care costs per capita. (See Exhibit 4.)
Even in Sweden, however, there is still room for considerable improvement. In 2009, BCG worked closely with a broad set of stakeholders there—payers, providers, industry, academia, and government—to propose a national, value-based strategy for using the registries to enhance health care delivery. The analysis showed that a $70 million annual investment in Sweden’s registries, data analysis resources, and IT infrastructure would, over ten years, generate a cumulative return of more than $7 billion. This tenfold payback would be achieved by reducing the annual growth in health care costs from an estimated 4.7 percent to 4.1 percent. The estimated economic value reflects the difference between the annual savings that could be achieved by measuring outcomes more systematically and the costs associated with setting up and running the registries.
- The savings were based on the assumption that measuring and sharing outcomes on a systematic basis would lower the cost of treating diseases by 1.5 percent per year. (Long-term studies of Swedish registries and other systems have demonstrated annual savings in the range of 1 to 3 percent.) To achieve these savings, Sweden would need to expand the coverage of its registries. BCG estimated that Sweden could more than double the proportion of health care expenditures covered by the registries, from 25 to 56 percent, by undertaking two initiatives: adding about 35 new registries and boosting participation in each registry to 85 percent of all patients with a given disease. Both initiatives could be accomplished in less than ten years. (The analysis assumed that the savings would gradually build to 1.5 percent per year for new registries.)
- The costs associated with setting up and running the registries were estimated on the basis of four components. Direct registry funding is the largest cost driver at an estimated $500,000 per registry per year. IT investments would also be significant, at $80 million over an eight-year period. Other costs include building competence centers ($25 million over four years) and running the program management office (PMO) and audit functions ($4 million per year).
Sweden’s march toward a value-based future is certainly not complete, but the country’s progress—along with the potential for substantial economic benefits—offers lessons for other health care systems. Above all, Sweden’s registries demonstrate that data on outcomes—when supplied and validated by clinicians and utilized to develop more effective treatment protocols and adjust them to specific patient populations—can unlock significant value by enabling better, more cost-effective care while eliminating unnecessary and ineffective treatments.
Putting VBHC into Practice
Comprehensive, authoritative registries are the backbone of the value-based approach. They provide the context for developing more effective treatment protocols. But while registries are a prerequisite, they are not by themselves sufficient. Several other elements are also important to the development of VBHC. Three elements, in particular, are critical to reordering the priorities of payers and providers.
Reimbursement. For a value-based approach to move from concept to reality, doctors and other medical staff, along with the institutions they work in, must be given incentives to produce better outcomes for patients. The same incentives would have an indirect effect on pharmaceutical and device suppliers, which would stand to gain by developing products and services that make a real difference rather than bringing to market “me too” products that may have little impact on improving outcomes in the treated population.
- Competition at the Right Level. Providers and suppliers alike need to compete on the basis of the delivery of value (patient outcomes relative to cost) rather than the price per procedure.
- Integrated Care. As with disease management, the delivery of care should be based on the patient’s condition. The system needs to promote treatment when a condition is more manageable—for example, via preventive measures—as well as when intervention is clearly required.
A number of countries are starting to build the infrastructure and processes to support a value-based approach. Countries that have national health care systems are making headway in systematizing the recording and collection of data. Other countries are piloting alternative reimbursement and incentive models that reward quality. On the whole, the conversion to a value-based approach is likely to be piecemeal and gradual, given the extent to which stakeholders are wedded to existing practices and wary of change.
To take hold, VBHC cannot be imposed by mandate—medical, commercial, and government stakeholders all need to be active and willing participants. The process is likely to involve negotiation, persuasion, and, above all, a clear demonstration of the advantages of a value-based approach. Nonetheless, stakeholders that take the lead in VBHC stand to gain a distinct competitive advantage by developing capabilities, expertise, and even specific products or services that are aligned with proven clinical outcomes. For example, a biopharma company that supports the development of registries and treatment protocols will be able to match its offerings to the practices promoted under a value-based system. This could lead to price premiums or more competitive, differentiated brand positions. It might also put the company in a better position to limit the risk associated with outcomes-based pricing agreements.
So who makes the first move? Each group of stakeholders has much to gain from a value-based approach, both independently and in collaboration with other stakeholders. The most obvious and immediate beneficiaries—apart from patients—are doctors, who would play a central role in developing and institutionalizing better treatment protocols. But both providers and payers have good reason to spearhead the development of a value-based approach—and for motives that are as much commercial as altruistic.
We are not blind to the barriers that must be overcome. But even a gradual shift to a VBHC system will lead to a focus on results rather than processes. Stakeholders will be rewarded on the basis of the value, rather than the volume, of the services they deliver to patients and payers. The internal contradictions of the present system will be replaced by a coherent framework in which stakeholders would, by design, be drawn into collaborative projects of mutual interest rather than zero-sum conflicts.
VBHC would also reenergize the provider industry by rewarding innovations that make a real difference and by engaging doctors and other medical staff in the progressive development and reform of the health care system. Other stakeholders will be able to mobilize their organizations with a renewed focus on innovation and improvement, all geared toward maximizing value.
Aside from all of these advantages—along with growing evidence that VBHC is not an idealized concept but a proven framework for reorganizing health care—perhaps the most compelling reason to adopt a value-based approach is the realization that the current system is unsustainable. Cost containment is not an effective strategy for dealing with the pressure on health care systems. Meanwhile, the uneasy standoff between steadily rising health care costs and mounting public debt will eventually reach a breaking point. Governments, payers, and providers cannot justify maintaining a system that does not align costs with outcomes. Preserving the status quo is as untenable as it is unethical.
Acknowledgments
First and foremost, the authors would like to thank the many entrepreneurial clinicians and registry leaders in Sweden who shared their experiences and data with us, as well as Carl Bennet of Getinge and Anders Ekblom of AstraZeneca, who cofounded the initial project with BCG, and senior leaders from Sweden’s health care system who engaged in the national strategy project in 2009. We would also like to thank Michael Porter and Jennifer Baron of Harvard Business School’s Institute for Strategy and Competitiveness for many stimulating discussions.
The authors are grateful to their BCG colleagues Simon Goodall, Jon Kaplan, Dave Matheson, Rasmus Molander, Johan Öberg, Andreas Ringman Uggla, and Peter Svensson for sharing their expertise and insights. The authors also acknowledge their colleagues who have contributed to BCG’s research on value-based health care, including Olga Berlinsky, Elena Bloudek, Anders Borg, Johan Ejerhed, Dan Grossman, Daniel Gorlin, Ann Keisu, Tarun Mahajan, Alexandra Martinsson, Rasmus Nerman, Mikael Nygård, Tobias Perdahl, Barry Rosenberg, Supriya Srinidhi, and Eric Wåhlgren.