Like the rest of the business world, biopharma and medtech companies are now entering the period of sustained uncertainty we call the “Fight” phase of COVID-19 response. During this phase—which could last 12 to 36 months, depending on when a vaccine is available—companies will continue to face heightened uncertainty, ongoing disruptions, and resulting impacts and changes to their operations. At the same time, operations leaders must start looking beyond these immediate challenges and prepare for the new reality that will follow.
Operations leaders must start looking beyond these immediate challenges and prepare for the new reality that will follow.
There is a very wide spectrum of potential outcomes over the medium term, and the right responses will vary depending on a company’s context and product segment—including generics, orthopedic medicines, personal protective equipment, life-saving biologics, and others. To help frame the issue, we have defined four broad-ranging scenarios for how the next several years could play out in the industry. By considering these scenarios in different contexts, we have identified a subset of actions that are universally applicable for virtually all biopharma and medtech companies:
- Anticipate and prepare for future disruptions.
- Accelerate investments to build digital and supply chain capabilities.
- Rethink the supply network.
- Embed flexibility and resilience.
Getting these actions right will not only help companies rebound from the current crisis—they will also help them prepare for the post-COVID future.
The Next 12 to 36 Months
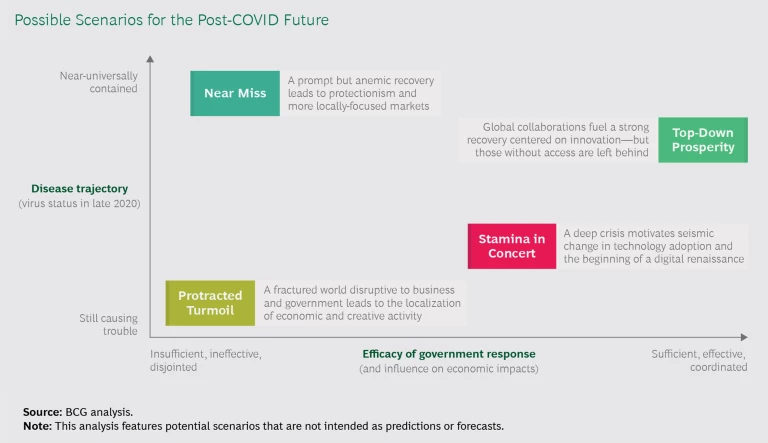
The degree of uncertainty in health care is unprecedented, making it hard for operations leaders to prepare for the future. Instead, they must prepare for a range of possible futures. Our work with biopharma and medtech operations teams has informed potential scenarios based on two primary factors: the trajectory of the coronavirus, and the efficacy of government responses. (See Exhibit 1.) Each of these macro scenarios would impact biopharma and medtech companies in different ways and, as a result, require different adjustments within operations functions. Of course, these scenarios are generalized and need to be built out for any specific company based on its portfolio, footprint, market exposure, and other factors.
Protracted Turmoil. If government actions to stem the spread of the coronavirus are insufficient, ineffective, and disjointed, and the virus continues to cause problems, the result would be a fractured world with ongoing business disruptions, reopenings and subsequent closures, and highly localized business activity. Isolationism would likely dampen trade dramatically over time, shortening supply chains and forcing global players to reevaluate their setups, potentially driven by significant government legislation or mandates. Some suppliers would almost certainly go bankrupt, as would many smaller hospital chains. Should this scenario play out, access to capital would be paramount.
Near Miss. Even if governments are unable to control the spread, the virus could ease on its own, leading to a prompt but anemic recovery characterized by increased protectionism and a market fragmented into fiefdoms. Regulatory frameworks would diverge, and companies would need to optimize their strategies for their most important markets. The incentive to innovate could diminish. The near-miss scenario would favor companies with strong balance sheets and access to key domestic markets, but it could also push many smaller players into bankruptcy.
Stamina in Concert. If the virus continues to cause problems, the world could still face a deep and prolonged crisis. Weakened economies would likely lead to pricing pressure and changes in care delivery (such as an increase in virtual interactions with patients and a decrease in elective procedures and visits). Along the same lines, the right government actions, bolstered by private investments, could spur a digital renaissance. Medtech and biopharma companies would adapt their supply chains by digitizing at an accelerated pace. And, as with prior scenarios, they would need to keep a close eye on protectionist policies, given the ongoing risk of disruption.
Top-Down Prosperity. Lastly, the combination of a contained virus and coherent, effective government policies could fuel a strong recovery centered on innovation. Protectionism would reverse, trade deals would return, and regulatory regimes would coalesce. An increase in consumer discretionary spending, coupled with a demand for health care innovation, would boost the overall industry. For health care companies, this scenario would mean the return of something close to the precrisis market—though a full return to previous conditions would remain unlikely. The industry would have a strong focus on innovation (in areas such as immunotherapies), and globalization would mean that supply chain management would remain a key driver of competitiveness worldwide. As with the prior scenario, investments in digital and proactive event planning would be critical.
Four No-Regret Moves
Each scenario has a corresponding set of actions that a company should take to prepare. Those actions will vary for each organization based on its product and service portfolio, operational footprint, supply chain setup, and many other elements. Although some actions will vary significantly, we believe there are four no-regret moves that will improve the competitiveness of all biopharma and medtech companies regardless of their context or which scenario comes to pass.
There are four no-regret moves that will improve the competitiveness of all biopharma and medtech companies regardless of their context or which scenario comes to pass.
Anticipate and prepare for future disruptions. First, all operations executives will need to strengthen their internal ability to plan for future dramatic changes in the market, factoring in their company’s market position, segment, and unique circumstances. Given that the coronavirus will likely not be the last major disruption we see in this generation, operations leaders should develop formal capabilities in event sensing, scenario planning, and reacting to future changes—not as an isolated exercise, but rather as an ongoing part of their strategic planning processes.
A best practice is to begin the process of “black swan planning” at regular intervals (typically quarterly), whereby the leadership team plays out a specific scenario, plans contingencies, and defines proactive actions to take. Some actions will be tied to how future events unfold; in those cases, operations leaders should determine which indicators they need to monitor and what levels represent threshold triggers for action. Companies should also develop specific implementation plans for those actions so they can move quickly as events unfold.
Companies will need to assign teams and resources for performing these scenario-planning activities and to coordinate effective end-to-end actions across the supply chain. These teams will need the right mandate, potentially reporting to the COO or supply chain head, and to closely link with strategic planning and business-continuity management processes. The teams must also include the right mix of operations experts, financial analysts, data scientists, and other specialists able to develop appropriate scenarios to test, along with recommended actions and monitoring mechanisms.
Accelerate investments to build digital and supply chain capabilities. During the immediate “Flatten” phase of the pandemic, we noticed a stark divide between companies that had mature digital capabilities and those that did not. The former were better equipped to take decisive, data-driven actions while the latter struggled to make sense of the limited data they had. As we move into the “Fight” phase, the uncertainties of the post-COVID world will persist, requiring greater supply chain resilience, responsiveness, and efficiency. Accordingly, medtech and biopharma companies should accelerate their investments in digital and supply chain capabilities, particularly those linked to predicting, identifying, and responding to disruptions in the most efficient and effective way. However, as most moving along the digital journey already know, real change requires more than the right analytics; it requires changes in business processes and behavior.
Historical patterns will no longer suffice to predict current or future demand.
The most foundational of the digital capabilities required is end-to-end visibility. Before the pandemic, biopharma and medtech companies had been investing in the ability to see the flow of materials across their supply chain and to understand the status and performance of their work centers in real time—often through a supply chain control tower. Today, biopharma and medtech companies will need to accelerate these investments and extend their scope well beyond their own assets. For example, they will need to extend visibility upstream to determine if any of their suppliers or inbound logistics providers are impacted by a disruption, and they must be able to respond accordingly. Similarly, they will need to understand how health care providers and patients are behaving in order to get an earlier read on how demand might be impacted by emerging events. Historical patterns will no longer suffice to predict current or future demand. Creating true end-to-end visibility will mean collaborating on a different level with supply chain partners and establishing partnerships with data providers that can enable more active risk monitoring.
Beyond visibility, biopharma and medtech companies will need to evaluate the impact of emerging disruptions and the potential benefits of mitigating actions in near real-time, so that they can adjust inventory and production processes to respond most effectively—particularly for products that are suddenly in high demand or where demand has disappeared. For example, companies will need to be able to decide whether they should buy more raw materials or secure greater capacity for a product based on very early information, even before the actual demand is impacted. Advanced analytics can support this type of decision making, but the data and analytics infrastructure must be in place before the need arises.
Although analytics are a critical part of the equation, COVID has truly tested the governance and decision making at many organizations. For example, sales- and operations-planning processes were often not able to respond to either dramatic declines in demand or extreme supply shortages. While it has been painful, the opportunity looking forward is to revisit core planning processes to see where thresholds were tested (or exposed), where the right stakeholder wasn’t involved at the right time, and where traditional ways of working became obsolete. There are various other capabilities to consider in concert, such as business continuity planning, setting company aspirations, and tailoring product supply strategies. Our experience thus far is that few companies would say their operating model worked optimally in response to the disruptions caused by the crisis.
Companies that thoughtfully determine where better visibility and analytics would have materially improved their response, and where their operating model showed cracks, will quickly develop a prioritized roadmap to build specific capabilities.
Rethink the supply network. Beyond broad scenario planning, medtech and biopharma companies will need to update their network strategy to ensure they are appropriately considering multiple dimensions of risk. Several possible scenarios and implications would require significant shifts to the network. For example, news media outlets are already discussing the potential for governments to mandate that companies localize the production of critical medicines and supplies as a response to the virus. Other contemplated measures would put restrictions on sourcing medical products or critical inputs from certain geographies. Even in the absence of government actions, firms could collaborate with pharma companies or CMOs to localize the production of certain products. These actions would force biopharma and medtech companies to reshape their production networks.
In addition, crisis-related disruptions exposed potential weaknesses in many companies’ supply chains, ranging from output reductions at short-staffed production plants, to logistics delays as key ports became backlogged, to delays in supplier deliveries of components or raw materials. Most companies avoided serious disruption to the supply of key products, but that experience has highlighted the sensitivity of many global supply chains to potential disruptions.
Although it is too early for biopharma and medtech companies to respond to these potential government actions, it is not too early for them to reevaluate risks in their current network design, nor to reset their network strategy approach to better prepare for future action. This is even more true given the long lead times for some of the potential moves that may be required.
Companies should accordingly begin to update the criteria they use to evaluate network actions to better incorporate considerations related to resilience and flexibility. In terms of resilience, companies should consider whether a particular move is likely to help them respond to a wider range of future scenarios. In terms of flexibility, companies should evaluate their ability to unwind or repurpose a particular investment if circumstances change, as well as their ability to access external capacity in different geographic locations. And, at least in the near term, companies need to consider potential legislation or government directives—and the resulting changes that would occur (as well as the investment that such changes would require).
To adapt their network strategy efficiently and effectively, companies will need to evolve beyond traditional modeling approaches and build up more dynamic capabilities, particularly in line with the supply chain measures discussed above. This will require not only investments in teams and technology, but also in upskilling leaders so that they can better understand and leverage the new capabilities to make decisions.
Embed flexibility and resilience. Like most organizations, biopharma and medtech companies are already adapting to COVID-related workplace disruptions, including changing operating procedures at their factories, implementing remote work for employees at headquarters, and exploring new technologies, such as contact tracing, to enable teams to return to work. However, companies have applied these measures in an ad hoc manner and inconsistently across teams and geographies. That approach—though to some extent unavoidable given the scope of uncertainty and the differences in government action across geographies—is not sustainable for the future.
Organizations must start preparing for an environment in which disruptions are the norm and not the exception—and develop ways of working that position the organization to thrive regardless. This is particularly true for biopharma and medtech companies, which operate in a highly regulated environment. Given an uncertain future, these organizations must evolve their ways of working to become more flexible and resilient.
Some changes to ways of working are less about responding to disruptions and more about improving baseline operations and efficiency.
Some changes to ways of working are less about responding to disruptions and more about improving baseline operations and efficiency. For example, companies should fully transition to electronic batch records and other digital tools at the site level, eliminating the need for manual steps such as physical signatures, wherever possible. Similarly, companies could permanently adopt remote working and digital collaboration for nonessential personnel in functions such as finance, planning, and logistics. This would minimize the number of people that need to be physically present at a site for normal operations, though it calls for digital upskilling and technology investments to maintain efficiency. Lastly, as companies continue to explore and adopt automation technologies—such as robotics and AI-based process control—those tools could create opportunities to limit the number of employees involved (or enable them to work at safe distances during normal operations).
Other changes relate to how biopharma and medtech companies can most effectively respond to disruptions. For example, many companies have found that more lightly staffed plants are as productive, if not more productive, than fully staffed facilities. Based on that insight, they could define, and work with regulators to approve, skeleton crews as part of their standard operations. They could also leverage advanced analytics to enhance their workforce-planning capabilities. Similarly, they can invest in more robust communications and employee-management mechanisms, including contact tracing, that can be deployed during crises.
Whether permanent or event-driven, these types of changes will require careful planning and execution to implement, both to generate value in the medium term and to help the organization prepare for the next disruption. Given the regulated environment in medtech and biopharma—and the resulting slow pace of change—leadership teams must start now.
For biopharma and medtech companies, the current period of uncertainty poses unprecedented challenges in planning operations, but at a macro level, we believe that the four scenarios discussed here provide good boundaries to inform planning. The right approach will call for different specific actions among individual companies—tailored to their unique context—but we believe that the four overarching actions will be valuable for all. We also believe that most of these actions are not new for most companies. Rather, they will require refocusing, reprioritizing, and enhancing efforts already underway or planned in order to prepare for the post-COVID world—however it evolves—and whatever disruption comes next.